What is the First Ionisation Energy:
- The first ionisation energy of an element can also be described as the energy charge per mole for the process:
- M(g) → M+(g) + e-
- The amount of energy required to remove an electron from an atom depends on the number of protons in the nuleus of the atom and on the electronic configuration of that atom.
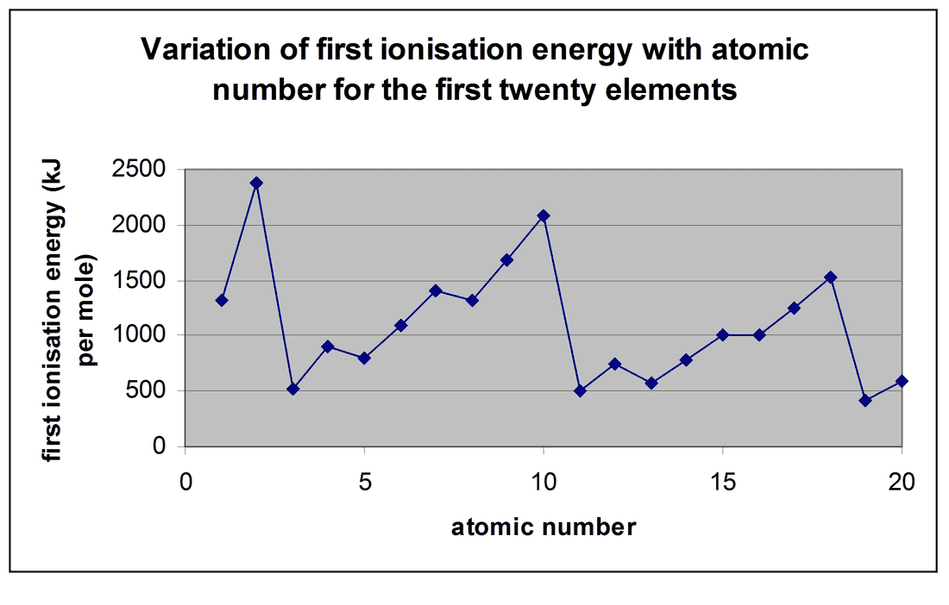
- The first ionisation energies of the first 20 elements in the periodic table is shown below:
- There are various trends in this graph which can be explainedby reference to the proton number and electronic configuration of the various elements. A number of factors must be considered:
- Energy is required to remove electrons from atoms in order to overcome their attraction to the nucleus. The greater the number of protons, the greater the attraction of the electrons to the nucleus and the harder it is to remove the electrons. The number of protons in the nucleus is known as the nuclear charge.
- The effect of this nuclear charge, however, is cancelled out to some extent by the other electrons in the atom. Each inner shell and inner sub-shell electron effectively cancels out one unit of charge from the nucleus. This is known as shielding.
- The outermost electrons in the atom thus only feel the residual positive charge after all inner shell and inner sub-shell electrons have cancelled out much of the nuclear charge. This residual positive charge is known as the effective nuclear charge.
- Electrons repel each other, particularly when they are in the same orbital. The degree of electron repulsion between the outermost electrons affecs the ease with which electrons can be moved.
- When considering trends in Ionisation energies, it is thus necessary to consider 4 factors:
- Nuclear Charge
- Shielding
- Effective Nuclear Charge
- Electron Repulsion