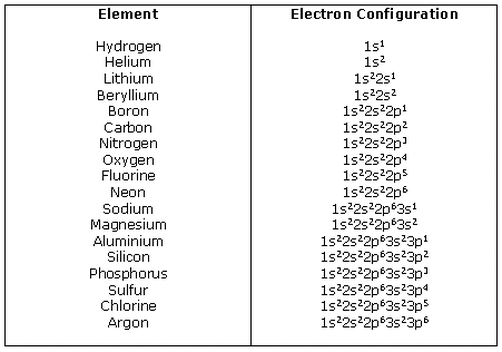
Electrons:
- Electrons repel each other.
- In a small space such as an orbital, it is impossible to put more than two electrons together.
- Since electrons are charged particles, and moving charges create a magentic field, it is possible to create a small magnetic attraction between two electrons if they are spinning in opposite directions in the same orbital. This is the reason two electrong, and not one, are permitted in the same orbital.
- It is thus possible to calculate the maximum possible number of electrons in each sub-level, and thus in each energy level:
Shell |
Number of electrons in each sub-level |
Maximum number of electrons |
1 |
2 x 1s |
2 |
2 |
2 x 2s, 6 x 2p |
8 |
3 |
2 x 3s, 6 x 3p |
8 |
4 |
2 x 4s, 10 x 3d, 6 x 4p |
18 |
5 |
2 x 5s, 10 x 4d, 6 x 5p |
18 |
6 |
2 x 6s, 14 x 4f, 10 x 5d, 6 x 6p |
32 |
Electron Arrangement in Orbitals:
- There are three rules which determine the way in which electrons fill the orbitals:
- Aufbau/Building Principle: Electrons always fill the lowest energy orbitals first.
- Hund's Rule: electrons never pair up in the same orbital until all orbitals of the same energy are singly occupied, and all unpaired electrons have parallel spin.
- Pauli Exclusion Principle: Only two electrons may occupy the same orbital, and they must do so with opposite spin.
- The arrangement of electrons in an atom is known as its electronic configuration. It can be represented in two ways:
- The arow and box method represents each orbital as a box and each electron as an arrow. The direction of the spin is shown by the orientation of the arrow.
- The orbital method indicates the number of electrons in eachorbital with a superscript written immediately after the orbital.
The Arrow and Box Method: |
The Orbital Method: |
- A shorthand form is often used for both the above methods. Full shells are not written in full but represented by the symbol of the noble gas element to which they correspond to, written in square brackets.
- EG: 1s²2s²2p^6 is represented as [Ne] and 1s²2s²2p^63s²3p^6 is represented as [Ar].
- The shorthand electronic configuration can be written as follows:
The Exceptions:
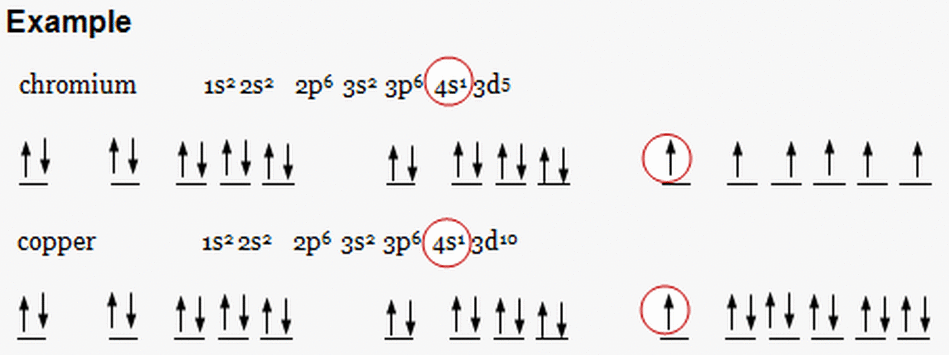
- The structures of copper and chromium are unusual based on the standard rules pplied to all the other atoms/elements.
- The difference in energy between the 3d and 4s electronsi very small, and in chromium the energy required to promote an electron from 4s to 3d is recovered in reduced repulsion which results from the fact that they are no longer paired. Thus the 4d^1, 3d^5 structure in Cr is preferred.
- In copper, the 3d orbitaks are actually lower in energy than the 4s orbital so the 4s^1, 3d^10 structure in Cu is preferred.
Electron Arrangement in Ions:
The electronic configuration of ions can be deduced by simply adding or removing the appropriate number of electrons. The order in which electrons are to be removed can be deduced from the following rules:
- Remove outer shell electrons first
- Remove p-electrons first, then s-electrons, then d-electrons
- Remove paired electrons before unpaired electrons in the same sub-level