The Second Ionisation Energy:
- M+(g) → M²+(g) + e-
The Third Ionisation Energy:
- M²+(g) → M³+(g) + e-
The Nth Ionisation Energy:
- M^(n-1)+(g) → M^(n)+(g) + e-
- It always becomes progressively more difficult to remove successive electrons from an atom; the second ionisation energy is always greater than the first, the third is always greater then the second, and so on.
- There are two satisfactory explanations for this: As more electrons are removed from an atom, the number of electrons remaining in the atom decreases. The repulsion between these electrons therefore decreases, while the number of protons remains the same. The remaining electrons are thus more stable and increasingly difficult to remove.
- The difference in successive ionisation energies, however, varies widely and depends on the electronic configuration of the atom in question.
- The difference in successive ionisation energies of an atom can be predicted qualitatively by consideration of the effective nuclear charge on the electron to be removed and the shielding of that electron by the inner shell and inner sub-shell electrons.
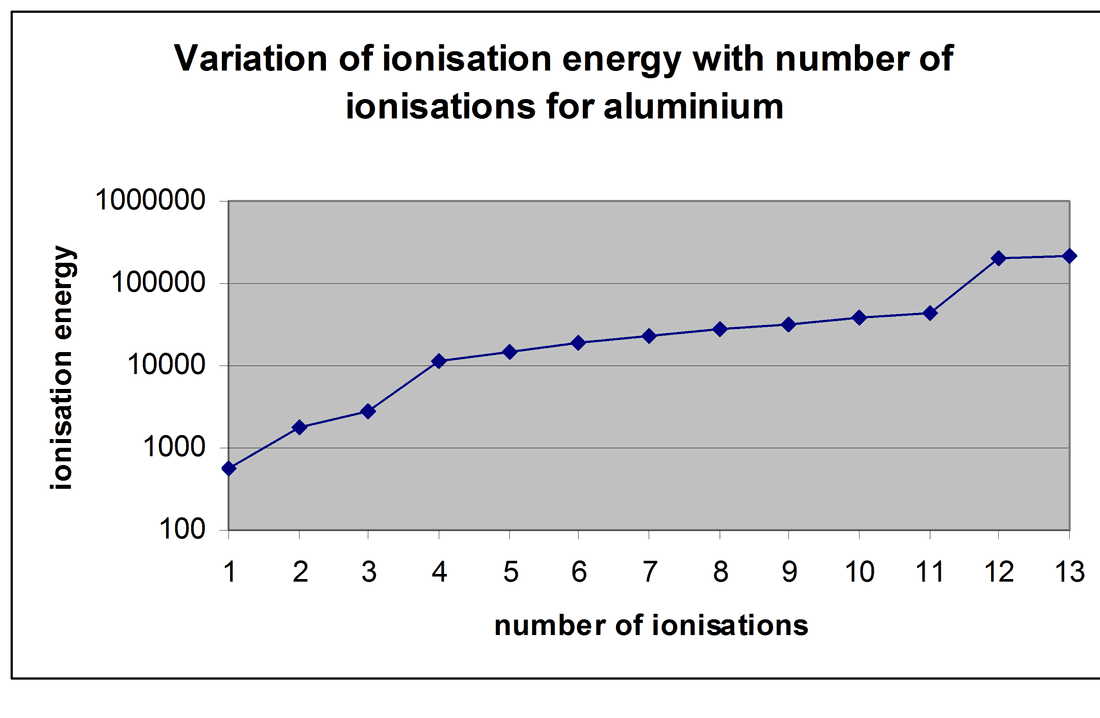
Successive Ionisation Energies of Aluminium(1s² 2s² 2p^6 3s² 3p¹):
IE |
1st |
2nd |
3rd |
4th |
5th |
6th |
7th |
8th |
9th |
10th |
11th |
12th |
13th |
Energy |
578 |
1817 |
2745 |
11578 |
14831 |
18378 |
23296 |
27460 |
31862 |
38458 |
42655 |
201276 |
222313 |
- 1st IE is fairly low because the 3 p electron is shielded by all the other electrons, and the effective nuclear charge is only +1.
- 2nd and 3rd IE are significantly higher than the 1st IE because 3s electrons are being removed and the effective nuclear charge on these electrons is +3.
- 1st: 578KJmol-1 2nd: 1817KJmol-1 3rd: 2745KJmol-1
- 4th IE holds a huge jump in ionisation energy, since a 2p electron is now being removed. The shielding has fallen and the effective nuclear charge has risen to +9.
- 5th and 6th IE are also high.
- 4th: 11578KJmol-1 5th: 14831KJmol-1 6th: 18378KJmol-1
- 7th IE holds another significant jump, since an unpaired 2p electron is now being removed.
- 7th: 23296KJmol-1 8th: 27460KJmol-1 9th: 31862KJmol-1
- 10th IE holds another significant jump, since a 2s electron is now being removed.
- 10th: 38458KJmol-1 11th: 42655KJmol-1
- 12th IE holds a huge jump, since a 1s electron is now being removed.
- 12th: 201276KJmol-1 13th: 222313KJmol-1
- These ionisation energies could then be plotted on a graph.